HOW LEMTRADA IS GIVEN AND MONITORED

Monthly Monitoring with LEMTRADA
LEMTRADA patient Donnie discusses how he made monthly monitoring a part of his treatment routine.
LEMTRADA IS AN INFUSION TREATMENT FOR RELAPSING MS.
LEMTRADA is given by intravenous (IV) infusion through a needle placed in your vein. When starting LEMTRADA, you will have 8 days of infusion, spread over two rounds of treatment that are about 12 months apart. Concurrent monitoring starts after your first infusion and continues until 4 years after the last round of treatment.
Additional infusion rounds and longer-term monitoring may occur if needed, as determined by your healthcare provider.
ROUND 1
- 1 infusion a day
- Given 5 days in a row
MONTHLY
Monthly monitoring for potential side effects starts upon completion of ROUND 1 and includes:
- monthly blood and urine tests
- self-checks
- yearly skin checks
ROUND 2
- 1 infusion a day
- Given 3 days in a row
ADDITIONAL ROUND(S)
- 1 infusion a day
- Given 3 days in a row
- Initiated no earlier than 1 year after your last treatment
MONTHLY
Monitoring will continue until 4 years or longer after the last round of treatment to help detect side effects like:
- autoimmune problems
- certain cancers
You will receive your infusions at a lab or healthcare provider’s office with equipment and staff trained to manage infusion reactions:
- It will take approximately 4 hours to receive 1 dose of LEMTRADA.
- You will be monitored closely during the infusion and for at least 2 hours following the completion of the infusion to watch for any infusion reactions.
- Be aware that administration time can vary, and you can expect to be at the infusion facility for up to 8 hours or more per day.
- LEMTRADA can cause serious side effects during infusion or up to 24 hours or longer after you receive LEMTRADA. Tell your healthcare provider immediately if you experience any discomfort during or after your infusion.

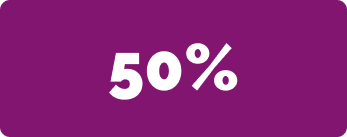
of patients did not require additional rounds of LEMTRADA or any other disease-modifying therapy (DMT)*†
*192 out of 387 patients
†Information beyond 3 treatment courses is limited
MONITORING IS JUST AS IMPORTANT AS GETTING TREATMENT.
Actual patient
Monthly monitoring to detect potential side effects starts after your first infusion and continues until 4 years or longer after the last round of treatment. You will need:
Monthly blood and urine tests

Ongoing symptom self-checks
Yearly skin exams
Certain side effects of LEMTRADA can happen months and even years after treatment. These can be, but are not limited to, autoimmune side effects and some kinds of cancers, including skin cancer (melanoma). That is why it is very important to have your blood and urine tested even if you’re feeling well and do not have any symptoms from LEMTRADA and relapsing MS.
Because of risks associated with autoimmune diseases, certain cancers, and infusion reactions, LEMTRADA is only available through a restricted program called the LEMTRADA Risk Evaluation and Mitigation Strategy (REMS). A REMS program is required by the FDA for certain medications to help ensure that the potential benefits of a drug outweigh its potential risks.
During an appointment, your healthcare provider will tell you more about the program and how to enroll.

Confirms patients are enrolled and educated about treatment and ongoing monitoring requirements.
Trains and certifies healthcare providers to prescribe LEMTRADA.
Verifies healthcare facilities are enrolled in the program and have on-site access to equipment and personnel trained to manage infusion reactions.
Makes sure approved pharmacies are certified with the program and working with trained facilities.
Important Safety Information
LEMTRADA is a prescription medicine used to treat relapsing forms of multiple sclerosis (MS), to include relapsing-remitting disease and active secondary progressive disease, in adults. Since treatment with LEMTRADA can increase your risk of getting certain conditions and diseases, LEMTRADA is generally prescribed for people who have tried 2 or more MS medicines that have not worked well enough. LEMTRADA is not recommended for use in patients with clinically isolated syndrome (CIS). It is not known if LEMTRADA is safe and effective for use in children under 17 years of age.
LEMTRADA can cause serious side effects including:
Serious autoimmune problems: Some people receiving LEMTRADA develop a condition where the immune cells in your body attack other cells or organs in the body (autoimmunity), which can be serious and may cause death. Serious autoimmune problems may include:
- Immune thrombocytopenic purpura (ITP), a condition of reduced platelet counts in your blood that can cause severe bleeding that may cause life‑threatening problems. Call your healthcare provider (HCP) right away if you have any of the following symptoms: easy bruising, bleeding from a cut that is hard to stop, coughing up blood, heavier menstrual periods than normal, bleeding from your gums or nose that is new or takes longer than usual to stop, small, scattered spots on your skin that are red, pink, or purple.
- Kidney problems called anti‑glomerular basement membrane disease, which, if not treated, can lead to severe kidney damage, kidney failure that needs dialysis, a kidney transplant, or death. Call your HCP right away if you have any of the following symptoms: swelling of your legs or feet, blood in the urine (red or tea‑colored urine), decrease in urine, fatigue, coughing up blood.
It is important for you to have blood and urine tests before you receive, while you are receiving and every month for 4 years or longer, after you receive your last LEMTRADA infusion.
Serious infusion reactions: LEMTRADA can cause serious infusion reactions that may cause death. Serious infusion reactions may happen while you receive, or up to 24 hours or longer after you receive LEMTRADA.
- You will receive your infusion at a healthcare facility with equipment and staff trained to manage infusion reactions, including serious allergic reactions, and urgent heart or breathing problems. You will be watched while you receive, and for 2 hours or longer after you receive, LEMTRADA. If a serious infusion reaction happens while you are receiving LEMTRADA, your infusion may be stopped.
Tell your HCP right away if you have any of the following symptoms of a serious infusion reaction during the infusion, and after you have left the healthcare facility:
|
|
To lower your chances of getting a serious infusion reaction, your HCP will give you a medicine called corticosteroids before your first 3 infusions of a treatment course. You may also be given other medicines before or after the infusion to try to reduce your chances of having these reactions or to treat them if they happen.
Stroke and tears in your arteries that supply blood to your brain (carotid and vertebral arteries): Some people have had serious and sometimes deadly strokes and tears in their carotid or vertebral arteries within 3 days of receiving LEMTRADA. Get help right away if you have any of the following symptoms that may be signs of a stroke or tears in your carotid or vertebral arteries: drooping of parts of your face, weakness on one side, sudden severe headache, difficulty with speech, neck pain.
Certain cancers: Receiving LEMTRADA may increase your chance of getting some kinds of cancers, including thyroid cancer, skin cancer (melanoma), and blood cancers called lymphoproliferative disorders and lymphoma. Call your HCP if you have the following symptoms that may be a sign of thyroid cancer: new lump, swelling in your neck, pain in front of neck, trouble swallowing or breathing, hoarseness or other voice changes that do not go away, cough that is not caused by a cold.
Have your skin checked before you start receiving LEMTRADA and each year while you are receiving treatment to monitor for symptoms of skin cancer.
Because of risks of autoimmunity, infusion reactions, and some kinds of cancers, LEMTRADA is only available through a restricted program called the LEMTRADA Risk Evaluation and Mitigation Strategy (REMS) Program.
Do not receive LEMTRADA if you:
- are allergic to alemtuzumab or to any of the inactive ingredients in LEMTRADA
- are infected with human immunodeficiency virus (HIV)
- have an active infection
Thyroid problems: Some patients taking LEMTRADA may get an overactive thyroid (hyperthyroidism) or an underactive thyroid (hypothyroidism). Call your HCP if you have: excessive sweating, unexplained weight loss, unexplained weight gain, fast heartbeat, eye swelling, nervousness, feeling cold, worsening tiredness, constipation.
Low blood counts (cytopenias): LEMTRADA may cause a decrease in some types of blood cells. Some people with these low blood counts have increased infections. Call your doctor right away if you have symptoms of cytopenias such as: weakness, chest pain, yellowing of the skin or whites of the eyes (jaundice), dark urine, fast heartbeat.
Inflammation of the liver: Call your HCP right away if you have symptoms such as unexplained nausea, stomach pain, tiredness, loss of appetite, yellowing of skin or whites of eyes, or bleeding or bruising more easily than normal.
Hemophagocytic lymphohistiocytosis: LEMTRADA may increase the risk of overactivity of the immune system that can be fatal if not diagnosed and treated early. If you experience symptoms such as fever, swollen glands, or skin rash, contact your HCP right away.
Adult Onset Still's Disease (AOSD): LEMTRADA may cause AOSD, a rare condition that can cause a high fever lasting more than 1 week, pain, stiffness with or without swelling in multiple joints, and/or a skin rash. If you experience a combination of these symptoms, contact your HCP immediately.
Thrombotic thrombocytopenic purpura (TTP): LEMTRADA may cause blood clotting problems that can be fatal. Call your HCP right away if you experience symptoms such as: purplish spots on skin or in mouth due to bleeding under skin, yellowing of skin or whites of eyes (jaundice), feel tired or weak, very pale skin, fever, fast heart rate or short of breath, headache, speech changes, confusion, vision changes, seizure, low amount of urine or dark or bloody urine, stomach pain, nausea, vomiting, or diarrhea.
Autoimmune encephalitis (AIE): LEMTRADA may cause AIE, a brain disorder which may include symptoms that seem like an MS relapse. Call your HCP right away if you have any of the following symptoms: personality changes, mood changes, seeing things that are not there (hallucinations), agitation, short term memory loss, confusion, movement disorders, or seizures.
Bleeding disorder (acquired hemophilia A): LEMTRADA may cause acquired hemophilia A. Call your HCP right away if you have any of the following symptoms: bruising, nose bleeds, bleeding from a cut that may take longer than usual to stop, painful or swollen joints, blood in urine, dark or bloody stools.
Inflammation of the colon (colitis): Tell your HCP if you have symptoms of colitis, such as diarrhea (loose stools) or more frequent bowel movements, stools that are black, tarry or have blood or mucous, or severe stomach-area pain or tenderness.
Serious infections: LEMTRADA may cause you to have a serious infection while you receive and after receiving a course of treatment. Serious infections may include:
- listeria. People who receive LEMTRADA have an increased chance of getting a bacterial infection called listeria, which can lead to significant complications or death. Avoid foods that may be a source of listeria or make sure foods are heated well.
- herpes viral infections. Some people taking LEMTRADA have an increased chance of getting herpes viral infections. Take medicines as prescribed by your HCP to reduce your chances of getting these infections.
- tuberculosis. Your HCP should check you for tuberculosis before you receive LEMTRADA.
- hepatitis. People who are at high risk of, or are carriers of, hepatitis B (HBV) or hepatitis C (HCV) may be at risk of irreversible liver damage.
These are not all the possible infections that could happen while on LEMTRADA. Call your HCP right away if you have symptoms of a serious infection such as fever or swollen glands. Talk to your HCP before you get vaccinations after receiving LEMTRADA. Certain vaccinations may increase your chances of getting infections.
Progressive multifocal leukoencephalopathy (PML): A rare brain infection that usually leads to death or severe disability has been reported with LEMTRADA. Symptoms of PML get worse over days to weeks. It is important that you call your doctor right away if you have any new or worsening medical problems that have lasted several days, including problems with thinking, eyesight, strength, balance, weakness on one side of your body, using your arms or legs.
Inflammation of the gallbladder without gallstones (acalculous cholecystitis): LEMTRADA may increase your chance of getting inflammation of the gallbladder without gallstones, a serious medical condition that can be life-threatening. Call your HCP right away if you have stomach pain or discomfort, fever, nausea, or are vomiting.
Swelling of lung tissue (pneumonitis): Some people have had swelling of the lung tissue while receiving LEMTRADA. Call your HCP right away if you have shortness of breath, cough, wheezing, chest pain or tightness, or are coughing up blood.
Before receiving LEMTRADA, tell your HCP if you:
- have bleeding, thyroid, or kidney problems
- have a recent history of infection
- are taking a medicine called Campath® (alemtuzumab)
- have received a live vaccine in the past 6 weeks before receiving LEMTRADA or plan to receive any live vaccines. Ask your HCP if you are not sure if your vaccine is a live vaccine.
- are pregnant or plan to become pregnant. LEMTRADA may harm your unborn baby. You should use birth control while receiving LEMTRADA and for 4 months after your course of treatment.
- are breastfeeding or plan to breastfeed. You and your HCP should decide if you should receive LEMTRADA or breastfeed.
Tell your HCP about all the medicines you take, including prescription and over‑the‑counter medicines, vitamins, and herbal supplements. LEMTRADA and other medicines may affect each other, causing side effects. Especially tell your HCP if you take medicines that increase your chance of getting infections, including medicines used to treat cancer or to control your immune system.
The most common side effects of LEMTRADA include:
|
|
|
Tell your HCP if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of LEMTRADA. You may report side effects to FDA at 1-800-FDA-1088.
Click here for full Prescribing Information including Serious Side Effects.
Click here for medication Guide including Serious Side Effects.
Important Safety Information
LEMTRADA is a prescription medicine used to treat relapsing forms of multiple sclerosis (MS), to include relapsing-remitting disease and active secondary progressive disease, in adults. Since treatment with LEMTRADA can increase your risk of getting certain conditions and diseases, LEMTRADA is generally prescribed for people who have tried 2 or more MS medicines that have not worked well enough. LEMTRADA is not recommended for use in patients with clinically isolated syndrome (CIS). It is not known if LEMTRADA is safe and effective for use in children under 17 years of age.
LEMTRADA can cause serious side effects including:
Serious autoimmune problems: Some people receiving LEMTRADA develop a condition where the immune cells in your body attack other cells or organs in the body (autoimmunity), which can be serious and may cause death. Serious autoimmune problems may include:
- Immune thrombocytopenic purpura (ITP), a condition of reduced platelet counts in your blood that can cause severe bleeding that may cause life‑threatening problems. Call your healthcare provider (HCP) right away if you have any of the following symptoms: easy bruising, bleeding from a cut that is hard to stop, coughing up blood, heavier menstrual periods than normal, bleeding from your gums or nose that is new or takes longer than usual to stop, small, scattered spots on your skin that are red, pink, or purple.
- Kidney problems called anti‑glomerular basement membrane disease, which, if not treated, can lead to severe kidney damage, kidney failure that needs dialysis, a kidney transplant, or death. Call your HCP right away if you have any of the following symptoms: swelling of your legs or feet, blood in the urine (red or tea‑colored urine), decrease in urine, fatigue, coughing up blood.
It is important for you to have blood and urine tests before you receive, while you are receiving and every month for 4 years or longer, after you receive your last LEMTRADA infusion.
Serious infusion reactions: LEMTRADA can cause serious infusion reactions that may cause death. Serious infusion reactions may happen while you receive, or up to 24 hours or longer after you receive LEMTRADA.
- You will receive your infusion at a healthcare facility with equipment and staff trained to manage infusion reactions, including serious allergic reactions, and urgent heart or breathing problems. You will be watched while you receive, and for 2 hours or longer after you receive, LEMTRADA. If a serious infusion reaction happens while you are receiving LEMTRADA, your infusion may be stopped.
Tell your HCP right away if you have any of the following symptoms of a serious infusion reaction during the infusion, and after you have left the healthcare facility:
|
|
To lower your chances of getting a serious infusion reaction, your HCP will give you a medicine called corticosteroids before your first 3 infusions of a treatment course. You may also be given other medicines before or after the infusion to try to reduce your chances of having these reactions or to treat them if they happen.
Stroke and tears in your arteries that supply blood to your brain (carotid and vertebral arteries): Some people have had serious and sometimes deadly strokes and tears in their carotid or vertebral arteries within 3 days of receiving LEMTRADA. Get help right away if you have any of the following symptoms that may be signs of a stroke or tears in your carotid or vertebral arteries: drooping of parts of your face, weakness on one side, sudden severe headache, difficulty with speech, neck pain.
Certain cancers: Receiving LEMTRADA may increase your chance of getting some kinds of cancers, including thyroid cancer, skin cancer (melanoma), and blood cancers called lymphoproliferative disorders and lymphoma. Call your HCP if you have the following symptoms that may be a sign of thyroid cancer: new lump, swelling in your neck, pain in front of neck, trouble swallowing or breathing, hoarseness or other voice changes that do not go away, cough that is not caused by a cold.
Have your skin checked before you start receiving LEMTRADA and each year while you are receiving treatment to monitor for symptoms of skin cancer.
Because of risks of autoimmunity, infusion reactions, and some kinds of cancers, LEMTRADA is only available through a restricted program called the LEMTRADA Risk Evaluation and Mitigation Strategy (REMS) Program.
Do not receive LEMTRADA if you:
- are allergic to alemtuzumab or to any of the inactive ingredients in LEMTRADA
- are infected with human immunodeficiency virus (HIV)
- have an active infection
Thyroid problems: Some patients taking LEMTRADA may get an overactive thyroid (hyperthyroidism) or an underactive thyroid (hypothyroidism). Call your HCP if you have: excessive sweating, unexplained weight loss, unexplained weight gain, fast heartbeat, eye swelling, nervousness, feeling cold, worsening tiredness, constipation.
Low blood counts (cytopenias): LEMTRADA may cause a decrease in some types of blood cells. Some people with these low blood counts have increased infections. Call your doctor right away if you have symptoms of cytopenias such as: weakness, chest pain, yellowing of the skin or whites of the eyes (jaundice), dark urine, fast heartbeat.
Inflammation of the liver: Call your HCP right away if you have symptoms such as unexplained nausea, stomach pain, tiredness, loss of appetite, yellowing of skin or whites of eyes, or bleeding or bruising more easily than normal.
Hemophagocytic lymphohistiocytosis: LEMTRADA may increase the risk of overactivity of the immune system that can be fatal if not diagnosed and treated early. If you experience symptoms such as fever, swollen glands, or skin rash, contact your HCP right away.
Adult Onset Still's Disease (AOSD): LEMTRADA may cause AOSD, a rare condition that can cause a high fever lasting more than 1 week, pain, stiffness with or without swelling in multiple joints, and/or a skin rash. If you experience a combination of these symptoms, contact your HCP immediately.
Thrombotic thrombocytopenic purpura (TTP): LEMTRADA may cause blood clotting problems that can be fatal. Call your HCP right away if you experience symptoms such as: purplish spots on skin or in mouth due to bleeding under skin, yellowing of skin or whites of eyes (jaundice), feel tired or weak, very pale skin, fever, fast heart rate or short of breath, headache, speech changes, confusion, vision changes, seizure, low amount of urine or dark or bloody urine, stomach pain, nausea, vomiting, or diarrhea.
Autoimmune encephalitis (AIE): LEMTRADA may cause AIE, a brain disorder which may include symptoms that seem like an MS relapse. Call your HCP right away if you have any of the following symptoms: personality changes, mood changes, seeing things that are not there (hallucinations), agitation, short term memory loss, confusion, movement disorders, or seizures.
Bleeding disorder (acquired hemophilia A): LEMTRADA may cause acquired hemophilia A. Call your HCP right away if you have any of the following symptoms: bruising, nose bleeds, bleeding from a cut that may take longer than usual to stop, painful or swollen joints, blood in urine, dark or bloody stools.
Inflammation of the colon (colitis): Tell your HCP if you have symptoms of colitis, such as diarrhea (loose stools) or more frequent bowel movements, stools that are black, tarry or have blood or mucous, or severe stomach-area pain or tenderness.
Serious infections: LEMTRADA may cause you to have a serious infection while you receive and after receiving a course of treatment. Serious infections may include:
- listeria. People who receive LEMTRADA have an increased chance of getting a bacterial infection called listeria, which can lead to significant complications or death. Avoid foods that may be a source of listeria or make sure foods are heated well.
- herpes viral infections. Some people taking LEMTRADA have an increased chance of getting herpes viral infections. Take medicines as prescribed by your HCP to reduce your chances of getting these infections.
- tuberculosis. Your HCP should check you for tuberculosis before you receive LEMTRADA.
- hepatitis. People who are at high risk of, or are carriers of, hepatitis B (HBV) or hepatitis C (HCV) may be at risk of irreversible liver damage.
These are not all the possible infections that could happen while on LEMTRADA. Call your HCP right away if you have symptoms of a serious infection such as fever or swollen glands. Talk to your HCP before you get vaccinations after receiving LEMTRADA. Certain vaccinations may increase your chances of getting infections.
Progressive multifocal leukoencephalopathy (PML): A rare brain infection that usually leads to death or severe disability has been reported with LEMTRADA. Symptoms of PML get worse over days to weeks. It is important that you call your doctor right away if you have any new or worsening medical problems that have lasted several days, including problems with thinking, eyesight, strength, balance, weakness on one side of your body, using your arms or legs.
Inflammation of the gallbladder without gallstones (acalculous cholecystitis): LEMTRADA may increase your chance of getting inflammation of the gallbladder without gallstones, a serious medical condition that can be life-threatening. Call your HCP right away if you have stomach pain or discomfort, fever, nausea, or are vomiting.
Swelling of lung tissue (pneumonitis): Some people have had swelling of the lung tissue while receiving LEMTRADA. Call your HCP right away if you have shortness of breath, cough, wheezing, chest pain or tightness, or are coughing up blood.
Before receiving LEMTRADA, tell your HCP if you:
- have bleeding, thyroid, or kidney problems
- have a recent history of infection
- are taking a medicine called Campath® (alemtuzumab)
- have received a live vaccine in the past 6 weeks before receiving LEMTRADA or plan to receive any live vaccines. Ask your HCP if you are not sure if your vaccine is a live vaccine.
- are pregnant or plan to become pregnant. LEMTRADA may harm your unborn baby. You should use birth control while receiving LEMTRADA and for 4 months after your course of treatment.
- are breastfeeding or plan to breastfeed. You and your HCP should decide if you should receive LEMTRADA or breastfeed.
Tell your HCP about all the medicines you take, including prescription and over‑the‑counter medicines, vitamins, and herbal supplements. LEMTRADA and other medicines may affect each other, causing side effects. Especially tell your HCP if you take medicines that increase your chance of getting infections, including medicines used to treat cancer or to control your immune system.
The most common side effects of LEMTRADA include:
|
|
|
Tell your HCP if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of LEMTRADA. You may report side effects to FDA at 1-800-FDA-1088.
Click here for full Prescribing Information including Serious Side Effects.
Click here for medication Guide including Serious Side Effects.
Important Safety Information
LEMTRADA is a prescription medicine used to treat relapsing forms of multiple sclerosis (MS), to include relapsing-remitting disease and active secondary progressive disease, in adults. Since treatment with LEMTRADA can increase your risk of getting certain conditions and diseases, LEMTRADA is generally prescribed for people who have tried 2 or more MS medicines that have not worked well enough. LEMTRADA is not recommended for use in patients with clinically isolated syndrome (CIS). It is not known if LEMTRADA is safe and effective for use in children under 17 years of age.
LEMTRADA can cause serious side effects including:
Serious autoimmune problems: Some people receiving LEMTRADA develop a condition where the immune cells in your body attack other cells or organs in the body (autoimmunity), which can be serious and may cause death. Serious autoimmune problems may include:
- Immune thrombocytopenic purpura (ITP), a condition of reduced platelet counts in your blood that can cause severe bleeding that may cause life‑threatening problems. Call your healthcare provider (HCP) right away if you have any of the following symptoms: easy bruising, bleeding from a cut that is hard to stop, coughing up blood, heavier menstrual periods than normal, bleeding from your gums or nose that is new or takes longer than usual to stop, small, scattered spots on your skin that are red, pink, or purple.
- Kidney problems called anti‑glomerular basement membrane disease, which, if not treated, can lead to severe kidney damage, kidney failure that needs dialysis, a kidney transplant, or death. Call your HCP right away if you have any of the following symptoms: swelling of your legs or feet, blood in the urine (red or tea‑colored urine), decrease in urine, fatigue, coughing up blood.
It is important for you to have blood and urine tests before you receive, while you are receiving and every month for 4 years or longer, after you receive your last LEMTRADA infusion.
Serious infusion reactions: LEMTRADA can cause serious infusion reactions that may cause death. Serious infusion reactions may happen while you receive, or up to 24 hours or longer after you receive LEMTRADA.
- You will receive your infusion at a healthcare facility with equipment and staff trained to manage infusion reactions, including serious allergic reactions, and urgent heart or breathing problems. You will be watched while you receive, and for 2 hours or longer after you receive, LEMTRADA. If a serious infusion reaction happens while you are receiving LEMTRADA, your infusion may be stopped.
Tell your HCP right away if you have any of the following symptoms of a serious infusion reaction during the infusion, and after you have left the healthcare facility:
|
|
To lower your chances of getting a serious infusion reaction, your HCP will give you a medicine called corticosteroids before your first 3 infusions of a treatment course. You may also be given other medicines before or after the infusion to try to reduce your chances of having these reactions or to treat them if they happen.
Stroke and tears in your arteries that supply blood to your brain (carotid and vertebral arteries): Some people have had serious and sometimes deadly strokes and tears in their carotid or vertebral arteries within 3 days of receiving LEMTRADA. Get help right away if you have any of the following symptoms that may be signs of a stroke or tears in your carotid or vertebral arteries: drooping of parts of your face, weakness on one side, sudden severe headache, difficulty with speech, neck pain.
Certain cancers: Receiving LEMTRADA may increase your chance of getting some kinds of cancers, including thyroid cancer, skin cancer (melanoma), and blood cancers called lymphoproliferative disorders and lymphoma. Call your HCP if you have the following symptoms that may be a sign of thyroid cancer: new lump, swelling in your neck, pain in front of neck, trouble swallowing or breathing, hoarseness or other voice changes that do not go away, cough that is not caused by a cold.
Have your skin checked before you start receiving LEMTRADA and each year while you are receiving treatment to monitor for symptoms of skin cancer.
Because of risks of autoimmunity, infusion reactions, and some kinds of cancers, LEMTRADA is only available through a restricted program called the LEMTRADA Risk Evaluation and Mitigation Strategy (REMS) Program.
Do not receive LEMTRADA if you:
- are allergic to alemtuzumab or to any of the inactive ingredients in LEMTRADA
- are infected with human immunodeficiency virus (HIV)
- have an active infection
Thyroid problems: Some patients taking LEMTRADA may get an overactive thyroid (hyperthyroidism) or an underactive thyroid (hypothyroidism). Call your HCP if you have: excessive sweating, unexplained weight loss, unexplained weight gain, fast heartbeat, eye swelling, nervousness, feeling cold, worsening tiredness, constipation.
Low blood counts (cytopenias): LEMTRADA may cause a decrease in some types of blood cells. Some people with these low blood counts have increased infections. Call your doctor right away if you have symptoms of cytopenias such as: weakness, chest pain, yellowing of the skin or whites of the eyes (jaundice), dark urine, fast heartbeat.
Inflammation of the liver: Call your HCP right away if you have symptoms such as unexplained nausea, stomach pain, tiredness, loss of appetite, yellowing of skin or whites of eyes, or bleeding or bruising more easily than normal.
Hemophagocytic lymphohistiocytosis: LEMTRADA may increase the risk of overactivity of the immune system that can be fatal if not diagnosed and treated early. If you experience symptoms such as fever, swollen glands, or skin rash, contact your HCP right away.
Adult Onset Still's Disease (AOSD): LEMTRADA may cause AOSD, a rare condition that can cause a high fever lasting more than 1 week, pain, stiffness with or without swelling in multiple joints, and/or a skin rash. If you experience a combination of these symptoms, contact your HCP immediately.
Thrombotic thrombocytopenic purpura (TTP): LEMTRADA may cause blood clotting problems that can be fatal. Call your HCP right away if you experience symptoms such as: purplish spots on skin or in mouth due to bleeding under skin, yellowing of skin or whites of eyes (jaundice), feel tired or weak, very pale skin, fever, fast heart rate or short of breath, headache, speech changes, confusion, vision changes, seizure, low amount of urine or dark or bloody urine, stomach pain, nausea, vomiting, or diarrhea.
Autoimmune encephalitis (AIE): LEMTRADA may cause AIE, a brain disorder which may include symptoms that seem like an MS relapse. Call your HCP right away if you have any of the following symptoms: personality changes, mood changes, seeing things that are not there (hallucinations), agitation, short term memory loss, confusion, movement disorders, or seizures.
Bleeding disorder (acquired hemophilia A): LEMTRADA may cause acquired hemophilia A. Call your HCP right away if you have any of the following symptoms: bruising, nose bleeds, bleeding from a cut that may take longer than usual to stop, painful or swollen joints, blood in urine, dark or bloody stools.
Inflammation of the colon (colitis): Tell your HCP if you have symptoms of colitis, such as diarrhea (loose stools) or more frequent bowel movements, stools that are black, tarry or have blood or mucous, or severe stomach-area pain or tenderness.
Serious infections: LEMTRADA may cause you to have a serious infection while you receive and after receiving a course of treatment. Serious infections may include:
- listeria. People who receive LEMTRADA have an increased chance of getting a bacterial infection called listeria, which can lead to significant complications or death. Avoid foods that may be a source of listeria or make sure foods are heated well.
- herpes viral infections. Some people taking LEMTRADA have an increased chance of getting herpes viral infections. Take medicines as prescribed by your HCP to reduce your chances of getting these infections.
- tuberculosis. Your HCP should check you for tuberculosis before you receive LEMTRADA.
- hepatitis. People who are at high risk of, or are carriers of, hepatitis B (HBV) or hepatitis C (HCV) may be at risk of irreversible liver damage.
These are not all the possible infections that could happen while on LEMTRADA. Call your HCP right away if you have symptoms of a serious infection such as fever or swollen glands. Talk to your HCP before you get vaccinations after receiving LEMTRADA. Certain vaccinations may increase your chances of getting infections.
Progressive multifocal leukoencephalopathy (PML): A rare brain infection that usually leads to death or severe disability has been reported with LEMTRADA. Symptoms of PML get worse over days to weeks. It is important that you call your doctor right away if you have any new or worsening medical problems that have lasted several days, including problems with thinking, eyesight, strength, balance, weakness on one side of your body, using your arms or legs.
Inflammation of the gallbladder without gallstones (acalculous cholecystitis): LEMTRADA may increase your chance of getting inflammation of the gallbladder without gallstones, a serious medical condition that can be life-threatening. Call your HCP right away if you have stomach pain or discomfort, fever, nausea, or are vomiting.
Swelling of lung tissue (pneumonitis): Some people have had swelling of the lung tissue while receiving LEMTRADA. Call your HCP right away if you have shortness of breath, cough, wheezing, chest pain or tightness, or are coughing up blood.
Before receiving LEMTRADA, tell your HCP if you:
- have bleeding, thyroid, or kidney problems
- have a recent history of infection
- are taking a medicine called Campath® (alemtuzumab)
- have received a live vaccine in the past 6 weeks before receiving LEMTRADA or plan to receive any live vaccines. Ask your HCP if you are not sure if your vaccine is a live vaccine.
- are pregnant or plan to become pregnant. LEMTRADA may harm your unborn baby. You should use birth control while receiving LEMTRADA and for 4 months after your course of treatment.
- are breastfeeding or plan to breastfeed. You and your HCP should decide if you should receive LEMTRADA or breastfeed.
Tell your HCP about all the medicines you take, including prescription and over‑the‑counter medicines, vitamins, and herbal supplements. LEMTRADA and other medicines may affect each other, causing side effects. Especially tell your HCP if you take medicines that increase your chance of getting infections, including medicines used to treat cancer or to control your immune system.
The most common side effects of LEMTRADA include:
|
|
|
Tell your HCP if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of LEMTRADA. You may report side effects to FDA at 1-800-FDA-1088.
Click here for full Prescribing Information including Serious Side Effects.
Click here for medication Guide including Serious Side Effects.